
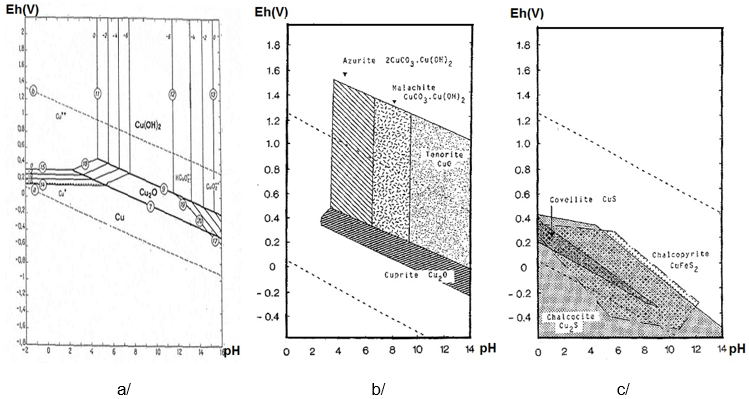
Despite the scarcity of available data on this subject, a comprehensive examination of the literature uncovered findings to the effect that the presence of acetate enhances the corrosion rate of copper 7 and that stress corrosion cracking can be induced in a-brass by sodium acetate 8. Despite exhaustive studies devoted to this subject with the purpose of elucidating the mechanism of passivity in sulphate 5 and chloride 6, very little information is available concerning the mechanism of copper dissolution in quasi-neutral aqueous solutions containing acetate anions. Electrodissolution of copper is a highly complex process and the composition of anodically formed layers depends on the nature of the electrolyte, the potential disturbance program, anodization time and mass transport 1-4. The corrosion resistance provided by a layer of reaction product varies according to the alloy and the composition of the electrolytic solution, and the thickness and electrical properties of a passive film are dependent on this composition. Although the initial corrosion rate of copper and its alloys may be high, it decreases as protective product layers are formed. Copper-nickel alloys are widely used in marine environments owing to their resistance to corrosion in saline solutions. The most important groups of copper alloys are brasses, bronzes, aluminum bronzes and cupronickels. Copper and copper alloys are widely used in practical applications due to their corrosion resistance and antifouling properties. The literature on the electrochemical behavior of copper in acidic and basic media is quite extensive, since many technological applications require knowledge of these processes. Keywords: acetate, copper/nickel, corrosion With increasing concentrations of acetate, the R P of the alloy and the Cu increased while that of the Ni electrode decreased. The impedance measurements taken at the open circuit potential revealed that the polarization resistance (R P) of the electrodes decreased in the following order: Ni > Alloy > Cu. Although the anodic current densities of Ni and the alloy increased with greater concentrations of acetate, the opposite effect occurred in Cu. The shapes of the potentiodynamic I(E) curves of the alloy were found to be quite similar to those of the Ni voltamograms. The data obtained from different electrochemical techniques were analyzed to determine the influence of Ni and Cu on the electrochemical processes of the alloy electrode. This paper discusses the electrochemical behavior of copper, nickel and a copper/nickel alloy in aerated aqueous 0.10 and 1.0 mol L -1 sodium acetate. Instituto de Química, Universidade Federal do Rio Grande do Sul,Į-mail: * apresentado no 14° CBECIMAT, Águas de São Pedro, Reinaldo Simões Gonçalves, Denise Schermann Azambuja*, Alzira Maria Serpa Lucho, Marlisa Piovesan Reche, Annelise M. Electrochemical Studies of Copper, Nickel and a Cu55/Ni45 Alloy in Aqueous Sodium Acetate
